Dr. Rajiv Bahl
Secretary to Government of India Department of Health Research & Director General, Indian Council of Medical Research

Director Desk
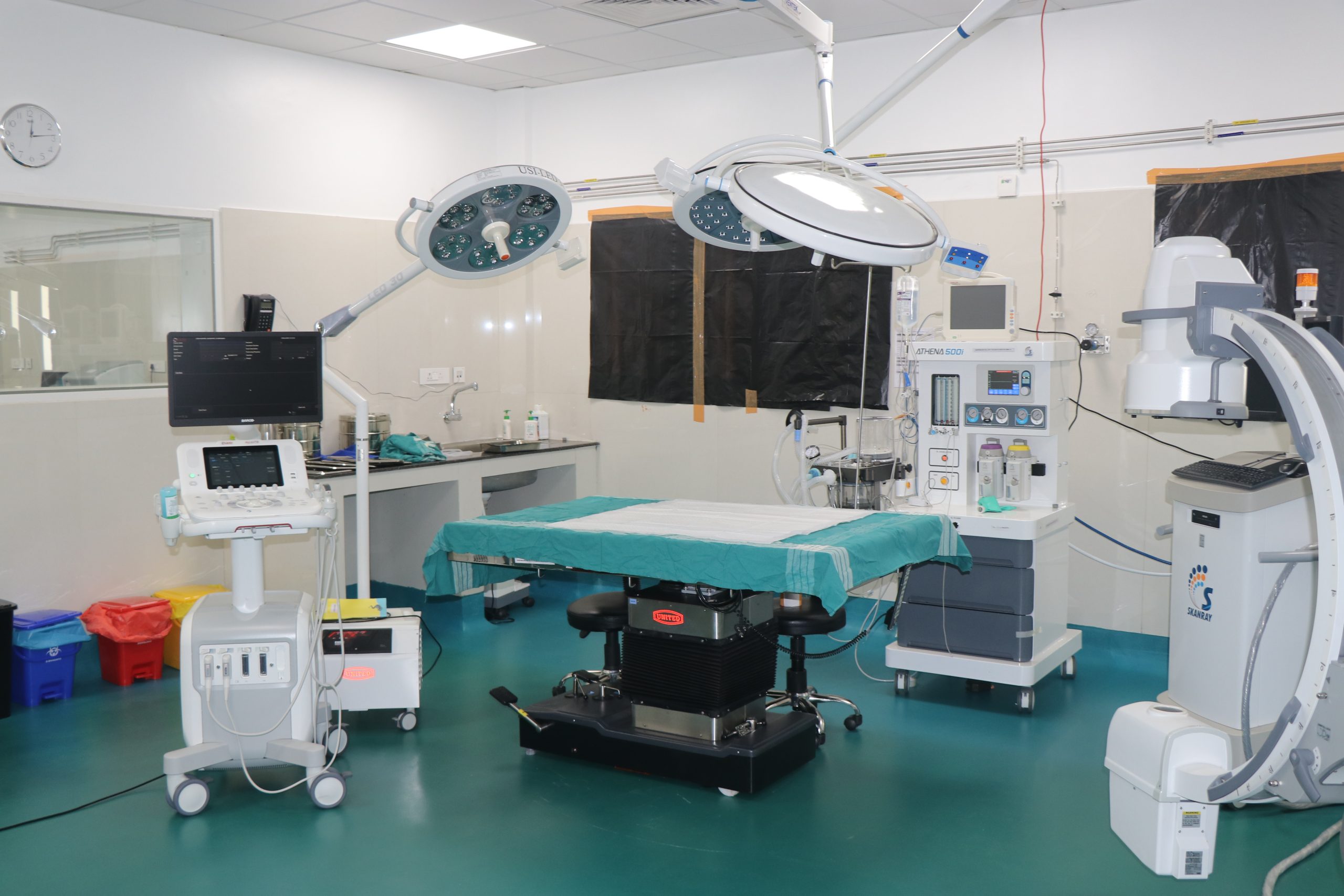
Pre-clinical trials on animals are crucial for developing new drugs, prophylactics, therapeutics, and medical devices. These trials are typically conducted before human clinical trials. They are vital for assessing safety, determining efficacy, understanding pharmacokinetics and pharmacodynamics, and exploring the mechanism of action of drugs, vaccines, and medical implants. Read More
 Read More
Read More The ICMR-NIPCR Film
The ICMR-NIPCR Film